The following expansion proceeds in one step against a constant pressure p2p2. In the limiting case, the value of the pressure pp abruptly changes from p1p1 and p2p2. 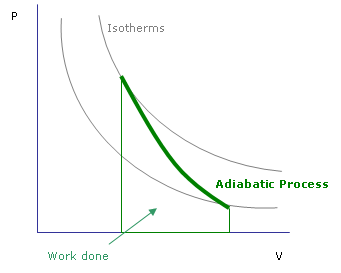
In contrast to the reversible expansion, an irreversible expansion is not performed continuously at equilibrium by means of infinitesimal changes in pressure. However, the values for the pressures p1p1 and p2p2 are given. The work done dW for the change in volume V by dV is given as PdV. Hence the work done is given by change in the internal energy of the gas for a closed system. The equation for an adiabatic process can be derived from the first law of thermodynamics relating to the change in internal energy dU to the work W done by the system and the heat dQ added to it. Let us first discuss the calculation of the work done during adiabatic expansion of gas, if in a closed system. Note that, in the given question, the volumes V1V1 and V2V2 are unknown. Adiabatic process is where no heat transfer occurs during the process. Where γγ is the ratio of the heat capacities γ=Cp/CV=Cm,p/Cm,V=cp/cVγ=Cp/CV=Cm,p/Cm,V=cp/cV. Since Cm,p−Cm,V=RCm,p−Cm,V=R, the exponent can be rewritten Where Cm,VCm,V is molar heat capacity at constant volume. Where $C_V$ is heat capacity at constant volume.Įquating $\mathrm dU$ and $\delta W$ yieldsĬ_V\,\mathrm dT
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
